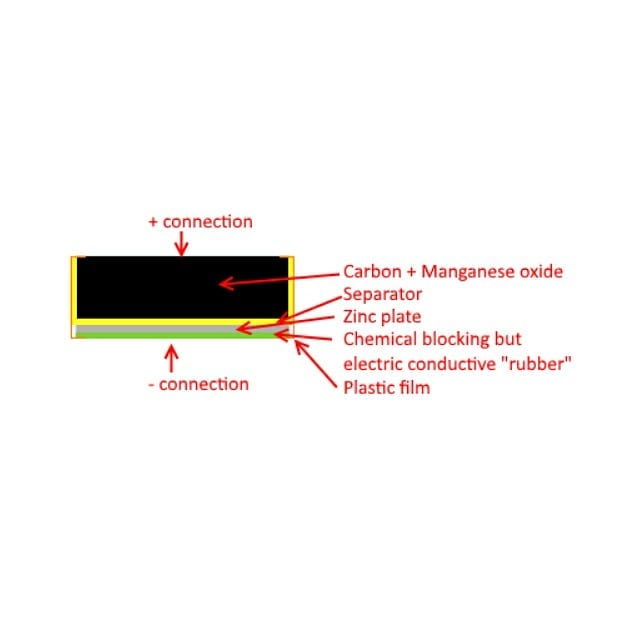
I was looking for a thin cylindrical battery for a project and thought of tearing down a 9V battery to repurpose its internal battery. To my surprise, I found a different stack inside it. I always thought (had opened one before) that these were made from six 1.5V cylindrical cells. This one seems to be a stack of six plastic pouches in series. A bit of googling helped me find that these are zinc-carbon battery pouches with a Zinc plate being the negative electrode, and the black brick (mixture of carbon + manganese oxide) being the positive electrode. Seems that manufacturers like Duracell go with the cylindrical cells and most others go with the pouch design. Learned something new today.
For those of you who do not know, most higher voltages in batteries are made by connecting smaller cells in series. Except for Lithium electrode batteries (which give cell voltages from 3V to 3.7V) most other known cell voltages range from 1.2V to 2V with usually 1.5V being the common one. This fundamental idea of increasing the overall battery voltage (by connecting in series) and increasing the battery capacity (by connecting in parallel) is the fundamental principle by which all batteries are made, ranging from the one powering your TV remote to the one in electric cars.



0 Comments
Comments are closed.